Description of the activity
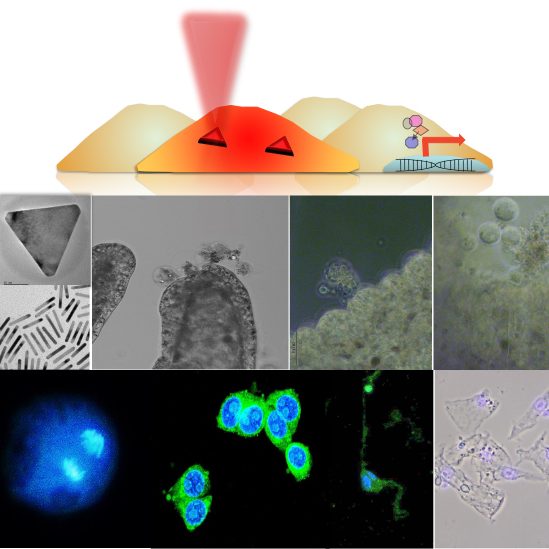
Hyperthermia is currently exploited as non-invasive approach to integrate conventional therapies, from cancer treatment to wound healing and tissue regeneration. Depending on the thermal shock intensity, cells exposed to high temperatures may activate programmed cell death pathways, undergo immediate necrosis or alternatively, in case of low heat, a mild hyperthermia may enhance wound healing. Heat-emitting nanoparticles that respond to externally applied forces represent fascinating tools for hyperthermia, allowing remote control of heat development, and in turn cell fate and decisions, for instance activating/inactivating specific pathways. In this context, we screened photothermal and magnetothermal properties of gold and magnetic nanoparticles in cell cultures and in vivo models, comparing the cell response to intracellular nano-sized heat sources (nanoheaters) to macroscopic heat sources. Following stimulation (alternating magnetic field for Fe3O4 NP and NIR laser for AuNP) the information extracted from Hydra, a model with unparalleled regenerative capacity, melanoma cells and human keratinocytes allow to screen nanoheaters performance inside living tissues (which may profoundly diverge from theoretical models) and suggest diverse biomedical applications ranging from cell ablation to tissue regeneration.
Involved personnel
A. Tino | C. Tortiglione | G. Tommasini | M. Rippa
National and International Collaborations
Instituto de Nanociencia y Materiales de Aragón (INMA), Zaragoza, Spain
Instrumentation/facilities
Advanced laboratory for molecular biology: equipment for DNA, RNA and Protein analysis; Cell culturing, animal facilities, fluorescence microscopy. Chemi doc, quantitative real Time PCR (StepOne, Applied Biosystem), cryostat, Functional Live Cell Imaging facility, Ultracentrifuge.
Diode laser system with emission in the near infrared region (λ = 1064 nm) controlled in current and temperature.
Active projects and contracts
ERC Starting grant 2019; European Research Council. Title: SIROCCO: Remote control of cellular signalling triggered by magnetic switching. Period of activity 2020-2025 (GA 8853468). Principal investigator: Maria Moros. ISASI scientific leader: C. Tortiglione
